Editors note: This post was originally published in September 2015 and has been completely revamped and updated for accuracy and comprehensiveness.
Are you suffering from any of the following symptoms: panic setting in when your phone’s battery is dying, frustration from not being able to find an open outlet at the airport, or being tangled in a web of charging cables? You just might be one of the billions experiencing inadequate battery life in smartphones.
Lithium-ion has dominated consumer electronics as the rechargeable battery of choice for years. These batteries are chosen by device manufactures for their high energy capacity and high cycle life, meaning they can be charged and discharged many times without degrading. However, lithium-ion technology hasn’t changed much in 20 years despite the shrinking and thinning form factor of mobile devices and the increase in complexity of software. There have been only marginal improvements to capacity, but manufacturers leverage it to power advanced chip and graphic specs instead of increasing device uptime.
Simply put, if you want more battery life out of a lithium-ion battery, you are going to have to purchase a larger device to house a larger battery. As long as the current materials for Li-ion batteries are used, there will not be a substantial improvement to battery life. The ceiling for battery life is only so high due to size/efficiency constraints of lithium-ion.
Looking towards the future, I will highlight emerging battery and charging advancements that will revolutionize battery efficiency.
Battery Technology
Lithium-Sulfur

Sulfur has long been used as an electrolyte in batteries due to its natural abundance on earth, but sulfur is not a great conductor and deteriorates easily. Researchers at both UT-Dallas and Penn State are now experimenting with adding molybdenum to the sulfur, which has shown to increase stability and conductivity to a point where it could be commercially viable.
These advanced compounds overcome the capacity ceiling of lithium-ion and are less expensive to make, weigh less, and are better for the environment. Initial testing has demonstrated 3x to 5x improvement in capacity as compared to Li-ion, and therefore 3x to 5x battery life in your smartphone. Coin batteries have been used for lab testing, however the compounds could be applied to smartphone batteries in the same manner.
Currently, issues with stability over many charge cycles need to be proven but the rising cost of materials to create traditional lithium-ion batteries and the high environmental impact will push for the advancement of Lithium-Sulfur (Li-S) sooner than later.
Graphene

Researchers at Samsung’s Advanced Institute of Technology (SAIT) have created a battery made of graphene “balls”. The synthesized graphene and silica, in the shape of spherical structures, are used as the anode and cathode in lithium-ion batteries. Lab testing has shown these batteries can be fully charged in as little as 12 minutes (5x faster) without risk of overheating.
While recharging batteries quicker is desired by consumers, this material does not yet increase the capacity of the Li-Ion battery and therefore does not affect battery life. Samsung has filed for a patent in both the US and Korea and we could see this rapid charging tech added to their consumer devices first.
Hydrogen Fuel Cell
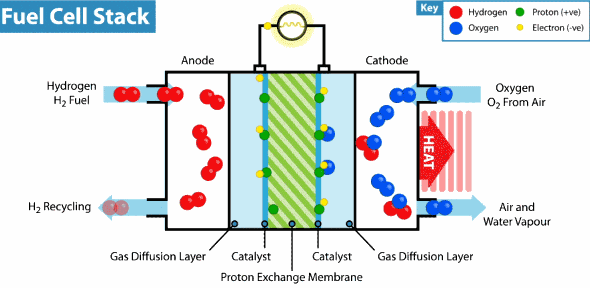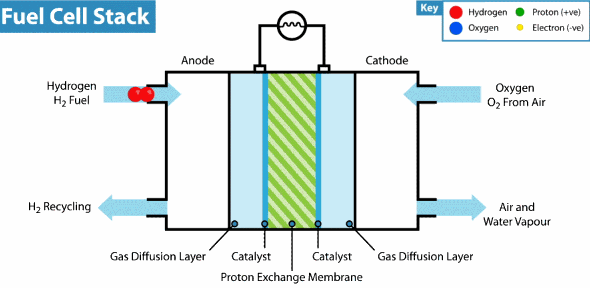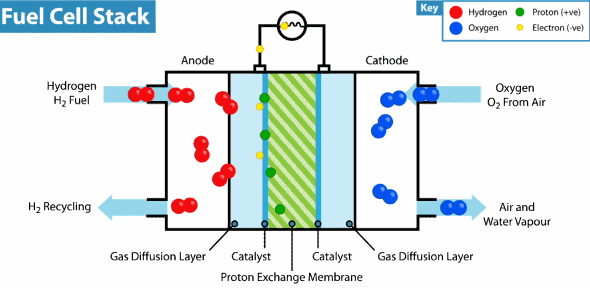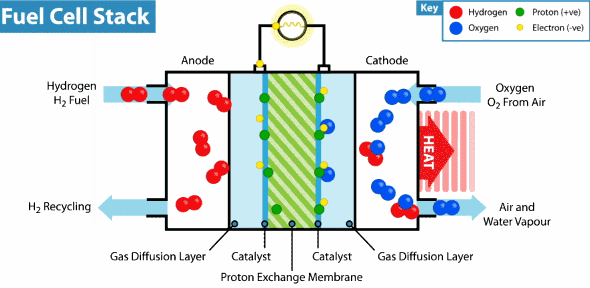
Intelligent Energy has developed a Hydrogen fuel cell that can provide a weeks worth of energy to a device with almost no impact on the environment. By combining hydrogen and oxygen in a thin fuel cell to generate power, there are no harmful chemicals or byproducts. In fact, the only emission from the battery is water vapor. Last month, Intelligent Energy installed a hydrogen fuel cell in an iPhone 6 without modifying the size or shape of the device. The only alteration was the addition of small rear vents to allow the water vapor to escape. They demonstrated that their new battery could almost seamlessly replace a lithium-ion one and increase battery life nearly seven fold. Recharging the battery requires refilling with clean Hydrogen and can be done in seconds.
Last year Intelligent Energy introduced an external, portable charger named Upp. This sunglasses case sized device will charge your phone up to 4 times before needing to be refilled with hydrogen. Swapping fuel cartridges will cost around $9 (though I couldn’t find any locations in BlueFletch’s hometown of Atlanta) and have no natural discharge over time. This makes Upp ideal for emergency preparedness, but the price point and lack of availability keeps it from being an everyday source of power.
Aluminum-Air
Aluminum-air batteries use oxygen naturally occurring in the air to charge their cathode. These batteries are incredibly lightweight and have already been tested on electric vehicles, providing 1,100 miles on a single charge. Unfortunately, these batteries must be replaced periodically and have a high production cost. If these two obstacles can be overcome, Aluminum-air batteries could provide multiple day battery usage at a fraction of the weight of lithium-ion.
Charging Technology
While device manufactures are waiting for the next proven battery technology to replace lithium-ion, there have been advancements in they ways we can charge our existing lithium-ion batteries.
Quick Charge 4+
Qualcomm has developed the Quick Charge technology currently available in over 150 Android devices. Using an intelligent algorithm, the device determines what power level to request at any point in time to allow optimum power transfer. The charge input is also routed via two different paths to optimize charging and reduce heat. With their latest 4+ version, Qualcomm claims a device can charge from dead to 50% in 15 minutes.
uBeam
A 25-year-old student developed wireless charging via ultrasound to transmit electricity. An adapter would need to be added to the device to receive the ultrasonic energy, but at only 5mm thick it can easily be concealed in a case or within the device. The transmitters are also only 5mm thick and could be placed nearly anywhere. This could mean that a phone is constantly charging via numerous transmitters encountered during the day.
Battery-free Technology

An interesting research project, looking even further into the future, is testing hardware that does not require a battery at all. Engineers at University of Washington have decoupled the analog and digital components in cellphones and placed the digital processors and radios separately (i.e. waist mounted or in a cell tower). Radio signals can then be bounced off the analog components to transfer analog audio. Successful phone calls and Skype calls have been completed from these devices and even video has been successfully transferred in the same way.
Conclusion
There have been many advancements and prototypes recently, but for the most part only third-party external batteries and chargers have been rolled out to consumers. In the coming years, expect to see manufactures develop their own batteries or partner with companies like the ones mentioned above. The final step to a truly wireless mobile phone is to eliminate the need to plug into a wall and charge a battery. With wireless charging advancements and new inventions in long-lasting battery technology, the next 5-10 years will bring weeklong battery life and unoccupied outlets at airports and coffee shops worldwide.